Number of Electrons Gained or Lost in Calcium
Carbon can not acquire or loseelectrons. How many electrons are lost or gained in the element Ca as it changes into simple ions.

Solved How Many Electrons Will An Atom Of Calcium Lose Or Chegg Com
Magnesium has a total of 12 electrons 2 in the innermost shell 8 in the second shell and two electrons in its valence shell third shell.

. Therefore the following atoms will lose or gain electrons as follows. Ca calcium has two electrons in its outer shell. State the number of electrons that must be gained or lost by an atom of calcium to achieve a stable electron configuration.
Use the atomic number to indicate the number of atoms of each type present in the compound. How many number of electrons gained or lost for calcium. Considering this How many electrons does fluorine gain or lose.
Calcium atoms have actually 20 electrons of which two are valence electrons. Calcium Express your answer as an ion. 5 Calcium Chlorine.
21 rows Total Number of Electrons in Neutral Atom Valence Electrons Gain or Lose Electrons Ion. In doing so they attain a noble gas electron configuration. Chemistry questions and answers.
In KCl both ions have full outer energy levels noble gas configuration. Express your answer using the term lost or gained. Number of Valence Electrons of electrons gained or lost to fill outer energy level.
Carbon is the nonmetal and with 4 valence electrons it will gain 4 electrons carbon gains and loses electrons depending on what the other element does in this case calcium wants to give electrons so carbon will gain electrons. The electronic configuration of calcium is 4s2. For example a neutral calcium atom with 20 protons and 20 electrons readily loses two electrons.
A fluorine atom can get a full valence shell by either gaining one more electron or by losing seven electronsThe former requires the transfer of less electrons so the fluorine atom will try to gain one electron first. Drag each tile to the correct box. Calcium atoms will lose two electrons in order to achieve the noble gas configuration of argon.
6 Magnesium Fluorine. State the number of electrons lost or gained when the following elements form ions. What number of electrons does calcium acquire or lose.
Calcium loses 2 electrons when it becomes an ion. Chemistry questions and answers. 2 electrons lost to go to the calcium ion.
During the formation of some compounds atoms gain or lose electrons and form electrically charged particles called ions link. When in profit one electron it also has the same number together argon. Therefore F ions are more common than F7 ions.
The atom gain or lose electron s to fulfil the octet rule. Consult the periodic table to help answer the question. Charge Oxidation Number Sodium.
A neutral calcium atom has 20 electrons while a calcium atom that has lost two electrons will have 18 electrons and a neutral argon atom also has 18 electrons. Sr Express your answer as an integer. Since calcium lost two electrons it has 20 protons but only 18 electrons.
When calcium atom loses electrons it becomes calcium ion. Fluorine atoms has 9 electrons and it has 7 valency electrons 2 7. Once a calcium atom loses 2 electrons it ends up v the same variety of electrons as argon i m sorry is a secure noble gas.
Electrons of Valence Electrons of Electrons Gained or Lost Oxidation Number Bromine Br 35 7 Gain 1 1-Lithium Li 3 1 Lose 1 1 Calcium Ca 20 2 Lose 2 2 Sulfur S 16 6 Gain 2 2-Boron B 5 3 Lose 3 3 Silicon Si 14 4 GainLose 4 4 4-Phosphorus P 15 5 Gain 3 3-2. Specify whether these 2 electrons were lost or gained by Sr. Carbon can not for C4-because If itpositive aspects 4 electrons it will be tough for thenucleus with six protons to maintain on to tenelectrons that is 4 additionalelectrons.
Number of electrons gained or lost in calciumhgtv dream home 2022 loft. Why carbon dont lose or acquire 4 electrons. Chlorine has 17 electron of i m sorry 7 room valence electrons.
The number of electrons present per shell in a calcium atom is 2 8 8 2. In this regard is calcium more likely to gain or lose electrons. Atoms of the main group elements either gain or lose electrons so they have eight electrons in the outermost energy level.
Does calcium lose or gain electrons. Calcium has two electrons in its outer most shell. For it to have a full valence level it will lose 2 electrons 2 8 8.
One element lose valence electrons and the atoms of another element gain valence electrons what are ionic bonds when nonmetal atoms group 1A2A3A share to a 4A5A6A7A8A share electrons to attain a noble gas arrangement. Calcium atoms will lose two electrons in order to achieve the noble gas configuration of argon. Calcium Ca Bromine Br lose 2 electrons gain 2 electrons Nitrogen N Krypton Kr lose 2 electrons about Oy Te.
When forming an ion those two electrons are lost and an. Select the number of electrons each atom needs to gain or lose to be consistent with the octet rule. Calcium has 2 valence electrons and is a metal so it will give up two electrons.
Match these elements with the number of electrons they gain or lose. April 10 2022. Using the model 2 8 8 2 Calcium atoms has 20 electron and it has 2 valency electrons 2 8 8 2.
A neutral calcium atom has 20 electrons while a calcium atom that has lost two electrons will have 18 electrons and a neutral argon atom also has 18 electrons. This is because calcium is in group two and so forms ions with a two positive charge. Does magnesium gain or lose electrons.
Gain 2 electrons lose 2 electrons gain 1 electron lose 1 electron. To complete it outer most shell and to become stable calcium atom losses two electrons.

Solved State The Number Of Electrons That Must Be Gained Or Chegg Com

3 433 Gostos 24 Comentarios Vanessa Adagio Studies No Instagram Managed To Get Stuff Done Today Notes Inspiration School Organization Notes Study Notes

Pin By Tiffanylynn20 On Schooldecor Study Notes Study Motivation Inspiration School Study Tips

How Many Valence Electrons Does Calcium Have
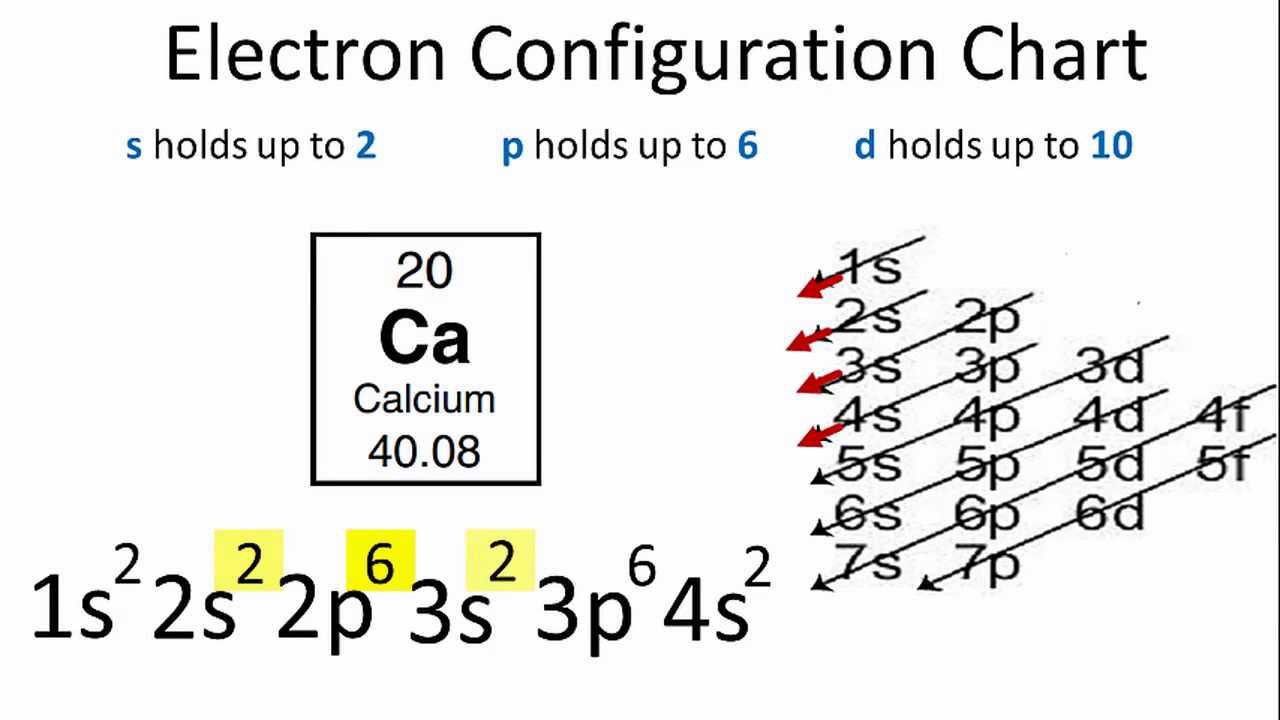
Electron Configuration For Calcium Ca

Draw The Lewis Structure Of Cabr2 Calcium Bromide Youtube

How Many Valence Electrons Does Calcium Have

Ca 2 Electron Configuration Calcium Ion Youtube
Valence Electrons Gaining And Losing Electrons Youtube

How To Determine Formula Of Calcium Chloride Equations Chemical Equation Writing

Structure Of Graphite In 2021 Covalent Bonding Electron Configuration Intermolecular Force

Quiz On Writing Chemical And Ionic Equations Equations Chemical Ionic
The Mass Of A Calcium Atom Is 40 Amu When It Loses Two Electrons To Become A Calcium Ion What Is Its New Mass Quora

Solved Calcium Atomic 20 Electron Configuration Of The Atom 2 Of Dots Around The Electron Dot Symbol Of The Atom Dots Number Of Electrons Lost Or Gained By The Atom To Form

Solved Question 2 Calcium Reacts With Bromine To Form Chegg Com

How To Determine Formula Of Calcium Carbonate Equations Chemical Equation Writing



Comments
Post a Comment